The Production and Properties of Devon Ball Clays
(A publication first produced by WBB in 1984 and subsequently updated )
1. Introduction

Fig. 1: Traditional clay cutting by hand in around 1920
The term ball clay is applied to plastic sedimentary clays, high in kaolinite, which possess white firing properties and high green strength. According to Scott (1) the name ball clay derives not from any specific property of the clay but from the original method of production, which consisted of cutting out the clay in open pits into cubes, the sides of which were approximately 22 to 25 cm, each weighing 14 to 16 kg. This original production method is illustrated in Fig. 1.
The ball clays of Devon have been in general use in pottery compositions in the British Isles since the latter half of the 17th century, and since the middle of the 19th century they have been exported to countries throughout Europe and to the United States. They are now marketed extensively throughout the world and owe their continuing popularity to their unique and consistent combination of physical properties.
The clays are won from two separate deposits. One, located between Bovey Tracey and Newton Abbot in South Devon, is known as ‘The Bovey Basin’; the other, located near Peters Marland, about 1.5 kilometres to the east of the village of Petrockstow in North Devon, is known as ‘The Petrockstow Basin’.
The geographical locations of both these deposits are shown in Fig.2.
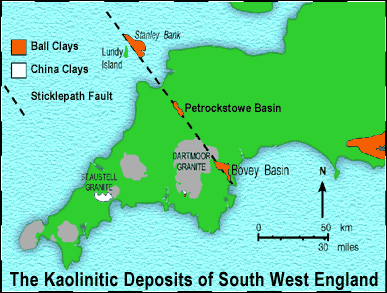 Fig. 2: The clay deposits of South West England
Fig. 2: The clay deposits of South West England
2. Geology
Most of the sedimentary rocks in Southwest England are of marine origin, excepting some terrestrial sands and conglomerates in the Crediton, Teignmouth and Exeter areas, and the lacustrine ball clay deposits near Newton Abbot and Torrington. To the west of Exeter the sedimentary rocks are dominated by slates and sandstones belonging to the Devonian and Carboniferous periods. These sediments range in age from 290 to 380 million years old.

Fig. 3: Mapping reserves and planning mining activity
The Devonian and Carboniferous marine sediments were involved in a major collision zone between two plates of continental crust some 280 million years ago. The sediments were folded and deformed, and then subsequently cut by large, northwest-southwest trending wrench fault systems; resulting in the melting of some parts of the continental crust and the intrusion of large volumes of granite magma. Granite now forms the central spine to the Southwest Peninsula, with older marine sediments draped over and around the granite areas of Dartmoor, Bodmin Moor, St Austell etc. (see Fig. 1)
Shallow water marine sediments continued to be deposited on the eastern side of the Peninsula until the Tertiary period (65 million years ago). By the middle of this period most of Southwest England was above sea-level due to the influence of the Alpine Mountain Building Period (Orogeny) in Central Europe. A further consequence of the Alpine Orogeny was the reactivation of the major northwest-southwest wrench fault systems in Southwest England. For the ball clays, the most important of these was the Sticklepath-Lustleigh fault system. This particular series of parallel faults extends from Torquay, across the eastern side of Dartmoor and North Devon, into the Bristol Channel near Lundy Island . (Fig. 2.)
The Sticklepath-Lustleigh faults played a major part in the deposition and preservation of the Tertiary ball clays in Devon.
Deposition and Derivation of the Ball Clays
The climate in the lower Tertiary period, in what is today Southwest England, was sub-tropical. The very warm, wet conditions resulted in the formation of a deep weathering mantle on both the Carboniferous and Devonian slates and the granite uplands and changed the feldspar minerals within the granite to a moderately ordered kaolinite. The unweathered slates have a clay mineralogy dominated by illite and chlorite. The weathering process converted all the chlorite and some illite to a disordered kaolinite. Bristow (1968) has demonstrated that the weathered Carboniferous slates preserved beneath the ball clays in North Devon show a complete gradation from fresh slate to ball clay mineralogies.
Reactivation and erosion along the Sticklepath-Lustleigh Fault System in the mid-Tertiary period resulted in a large northwest-southeast trending depression. Sediments derived from the erosion of the thick weathering mantle were brought in by the river systems. The coarser sands and gravels were deposited as alluvial sheets or fans on the valley floor and the clay and silt fraction and vegetation were deposited in shallow lakes. Large rafts of transported vegetation debris accumulated in the swampy lakes (Vincent 1974). Occasional drier interludes allowed the lakes to dry out and vegetation to spread across the exposed mudflats. These drier periods can be recognised by the presence of rootlet horizons and occasional silt filled sun-cracks in the ball clay seams.
The en-echelon configuration of the wrench fault system resulted in the formation of a series of tectonic pull-apart basins along the fault-line (see Fig. 1). Of these, the South Devon Bovey Basin and the North Devon Petrockstow Basin remain on land, and a third, the Stanley Banks Basin, has been identified in the Bristol Channel to the east of Lundy Island (Brooks and James 1975, Fletcher 1975). These tectonic basins were areas of subsidence which enabled the slow accumulation of ball clays, unconsolidated sands and lignites. Subsidence of the valley floor in the basin areas was probably linked to periods of earthquake activity and movement along the fault system. Several researchers have examined the formation and development of the basins (Bristow and Hughes 1971, Bristow et al 1992, Edwards 1976, Holloway and Chadwick 1986).
In the Bovey Basin a gravity survey recorded a maximum depth in excess of 1300 metres (Fasham 1971); at Petrockstow the British Geological Survey confirmed a maximum depth of approximately 750 metres (Freshney et al. 1979). These sediments have been dated by examining pollen samples and they range from upper Eocene to Middle Oligocene (35 to 28 million years old) (Chandler 1957, 1964).
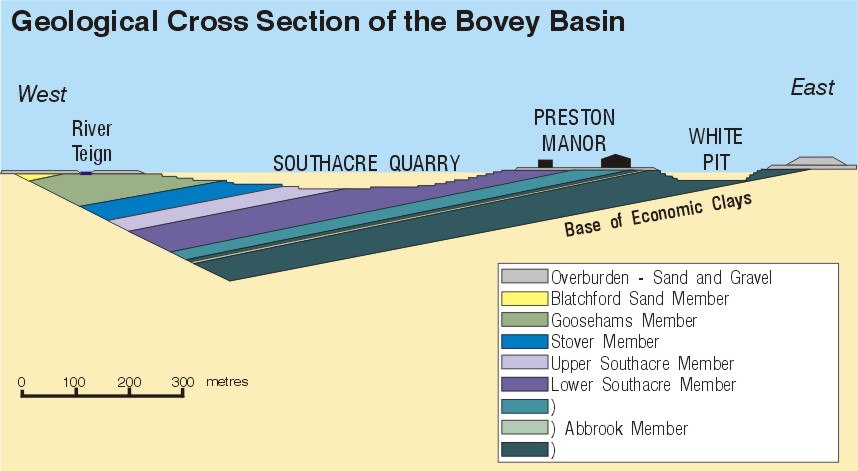
Fig. 4: Geological section across the eastern part of the Bovey Basin
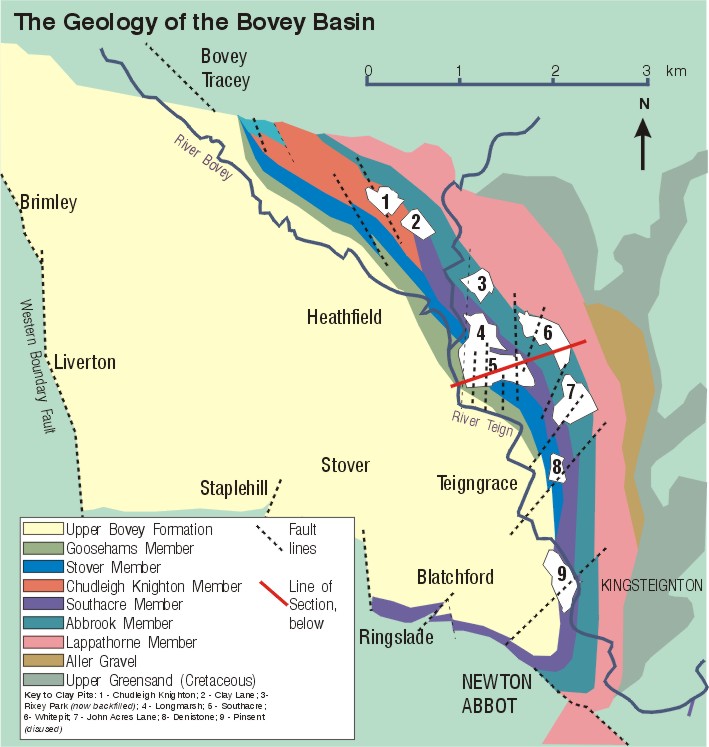
Fig. 5: The Bovey Basin
The South Devon Bovey Basin
The Bovey Basin extends for approximately 11km northwest – southeast and 6km in an east-west direction (see Fig. 5). Vincent (1974) and Edwards (1976) have recognised a complex sedimentary history, with sediments derived from different source areas. The ball clay mineralogy comprises varying proportions of disordered or ordered kaolinite, illite and quartz. The ball clays containing a high content of ordered kaolinite were largely derived from weathered granite. Those containing disordered kaolinite and illite are probably derived from weathered country rocks. Ball clays such as the Parkes seam, which largely consists of ordered kaolinite, was probably derived from kaolinised granite.
The strata within the basin are folded into a shallow syncline with its axis running northwest-southeast. The eastern limb of the syncline contains the majority of the opencast quarries, and bedding dips generally at 15 degrees to the southwest (see Fig. 4). Small scale, high-angle normal and reverse faulting affects some parts of the clay sequence.
The Chudleigh Knighton Member occurs in the northeastern part of the basin and contains clays with a high content of ordered kaolinite. There are some similarities with the Group 1 clays of the Southacre Member. Although carbonaceous in the south, the Member becomes sandier when traced northwards. This is the result of the sediment being derived from the northwest and being laid down in outwash fan-lake-backswamp
series of environments (Vincent 1974).
In the more medium-ordered kaolinite Southacre Member, this pattern is reversed and sand dominates the southern area, whilst to the north the Member becomes increasingly dominated by clays and lignites, so that the thickest lignite sequence occurs in the Twinyeo area, where the two backswamp phases were superimposed.
The underlying Abbrook Member contains more disordered kaolinite seams of Groups 2,3 and 4 intercalated with sands, but only minor lignite and carbonaceus clay.
The overlying Goosehams Member forms the top of the worked sequence in the eastern part of the basin. The clays are variably carbonaceous and in part resemble those found in the Southacre Member, but there can be a higher free quartz and illite content and occasional sand horizons. The Goosehams Member contains both Groups 1 and 2 clays. (See Figs. 4 & 5).
The Members forming the upper parts of the Bovey Formation occur in the central, western and northern parts of the basin. They comprise a series of sands, sandy clays and ball clays which are not currently worked.
The North Devon Petrockstow Basin
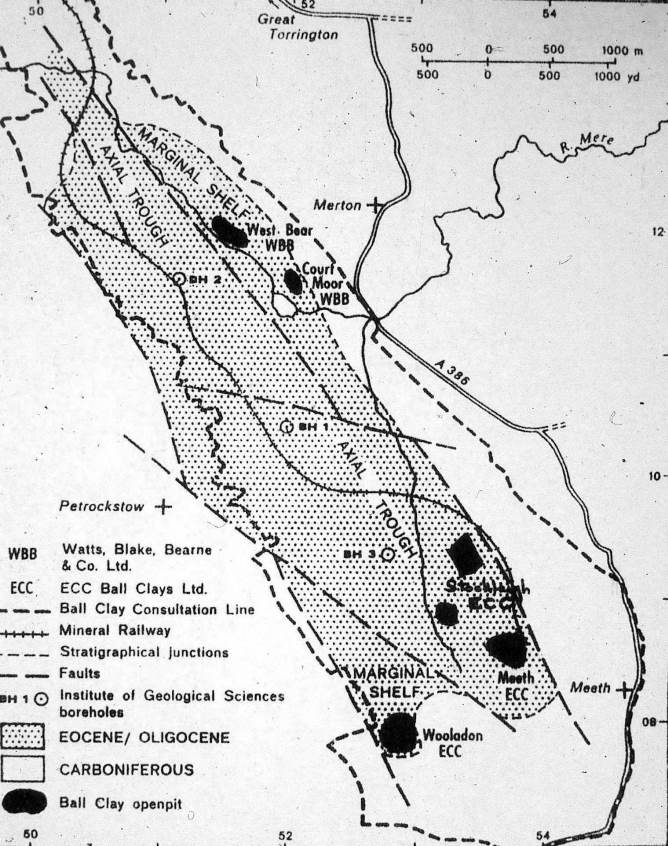 Fig. 6: The Petrockstow Basin
Fig. 6: The Petrockstow Basin
The Petrockstow Basin is narrower and more elongate than the Bovey Basin – being 6km northwest-southeast by 1.5km east-west. The basin is divided into a deep, axial trough and shallower, marginal shelves in the northeast and southern areas, which have been developed for clay extraction.
The northern-eastern shelf yields a sequence of plastic ball clays, sandy clays and carbonaceous clays with occasional sands and lignites. Nine separate sedimentary cycles or repetitions have been recognised in this area (Freshney et al 1979). The ball clays contain very disordered kaolinite and illite, indicating that weathered Carboniferous slate was the probable source material. The clays are generally less carbonaceous, stronger and more plastic than those extracted from the Bovey Basin.
3. Production
Ball clay is extracted predominantly from opencast workings. However, a small quantity of high quality clay is still extracted from underground mines in cases where surface access is impracticable and where higher production costs can be justified through exploitation of quality reserves. Nevertheless, as a result of economic considerations, the role of underground mining continues to diminish.
Shaft mining of ball clay began around 1870 and involved sinking vertical shafts to a maximum legal and practical depth of 50 metres. This method was used up until about 1960 and was subsequently replaced by inclined shafts, known locally as adits, which allowed access to underground mines.
Driven from the surface outcrop these adits followed the full-dip of a specific clay seam. Predominantly around 1000 metres in inclined length, these main tunnels provided access for men, materials and services to quality clays some 150 to 200 metres below the surface. Side tunnels following the strike of the seam were driven at right angles to the main adit and the clay was extracted by hand-held compressed air spaders or by electro-hydraulic cutter-loader machines. Small rail trucks transported the clay to surface storage sheds.
The last underground ball clay adit mine in Devon closed in South Devon in 1999.

Fig. 7: Cutter/loader machine at the clay face of an adit mine.
In opencast working, the exposed clay is won mechanically by means of hydraulic excavators, then loaded into dumpers and transported to the storage bays.
Throughout the extraction process the type and quality of the clay is monitored by Quarry Operators, in co-operation with the Works Quality Control Laboratory. At any one time the mining of some 40 to 50 different seams of clay may be taking place in various parts of the Bovey Basin, necessitating an equivalent number of storage bays. At Petrockstow, in North Devon, there are 12 to 15 storage bays, but thinner multiple seams of each selection are worked in the cyclic sediments of the two main open pits.

Fig.8: Clay passing through a static shredding machine.
The first stage in the process of bulk ball clay is size reduction and homogenisation. Bulk clays are stored in discrete bays according to quality type, which is determined by regular and comprehensive testing of samples drawn from the working faces and storage bays. Bulk clays are weight loaded from individual bays into tipper lorries, according to a blend recipe determined daily by Quality Control. This bulk blend is transported to a static shredding machine, where size reduction to around 25 millimetres and thorough homogenisation of individual clays is achieved to produce a shredded blend (Fig.8). Continual sampling of the clays during the shredding process is achieved by in-line automatic samples.
As with most naturally occurring minerals, variations in properties are not uncommon in ball clays. Because of their fine particle size, associated natural contaminants and, to some extent, their relatively low selling price, it was not until recently that economically viable methods for their purification and refining were developed.
Even today therefore, strict selection procedures at the pit face and frequent sampling and testing of the clays throughout the production process are essential (Fig. 9).
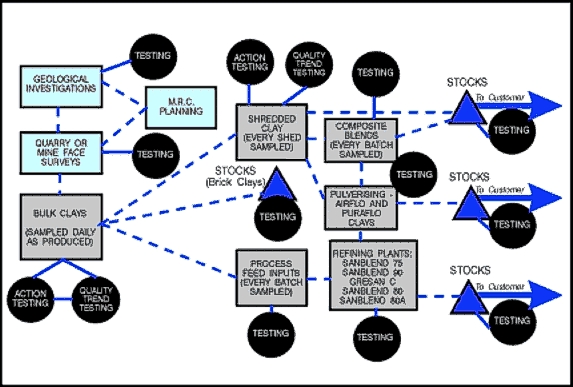 Fig. 9: Quality control testing during ball clay production
Fig. 9: Quality control testing during ball clay production
4. Processing

Fig.10: Modern opencast ball clay quarrying in South Devon.
Although most of the ball clay sold to the European ceramic industries is supplied in shredded form, considerable quantities are sold in pulverised air-floated form to other industries, as well as to ceramic producers in more distant locations. In certain cases for specialised uses, the clay may also have some additional form of chemical modification or treatment. The majority of the clay processed in this manner is sold in bagged, palletised form, but for deliveries within the United Kingdom, bulk transport by road tanker can be used.
Exports of ball clay to Northern Europe and the Mediterranean are made from the local port of Teignmouth (see Fig.12). Approximately 220,000 tonnes of ball clay are exported annually through this port. Exports to more distant countries are made through the deep water ports of Liverpool, Southampton, Ipswich and Felixstowe. Plate 5 shows the process of bagging and palletising powdered clay.

Fig 11: Bagging and palletising powdered clay

Fig 12: Loading at Teignmouth Docks
5. Refining
In 1975, Watts Blake Bearne & Co. produced the worlds first refined ball clay. This was Sanblend 75 – specially developed for use in the vitreous china sanitaryware industry. SanBlend 75 is a blend of both carbonaceous and non-carbonaceous clays, produced in noodle form in a purpose-built £2.5 million plant situated in South Devon. To produce SanBlend 75, carbonaceous clays undergo wet refining (being blunged with deflocculants), and coarse particles are removed by screening. Simultaneously, non-carbonaceous clays (of similar basic mineralogy) are dried and powdered and then air classified to remove coarse material. These two feedstocks are blended together with a non-carbonaceous thixotropic clay to form a plastic mass, and then noodled and dried to approximately 15% moisture.
Usage of refined clays makes production easier for ceramic manufacturers. Blunging time is considerably reduced, and the resulting slip is more consistent, homogeneous and stable. In addition, the negligible screen residue means less wastage and a reduction in screen cleaning requirements.
A second fully refined noodled ball clay blend, SanBlend GC, followed. This was specially formulated for gres sanitaire slip casting to provide a good fired colour and, if an appropriate chamotte is used, to obviate the need for an engobe. During the production process, each component clay is refined, so that in the final product a minimum of material coarser than 100 microns is present. In order to assist dispersion, SanBlend GC (like SanBlend 75) is pre-deflocculated during production. This not only assists blunging and slip preparation, but also provides an extremely high degree of stability and predictability to the resultant casting slip.
The most recent addition to the range is SanBlend 90, produced in a new plant, which was commissioned in 1995. SanBlend 90 combines three types of clay: a Carbonaceous blend, a low carbon and plastic clay blend, and a further low carbon blend. This fully-refined product is specifically designed for pressure-casting and fast-firing manufacture of sanitaryware, although it is also suitable for a range of other techniques.
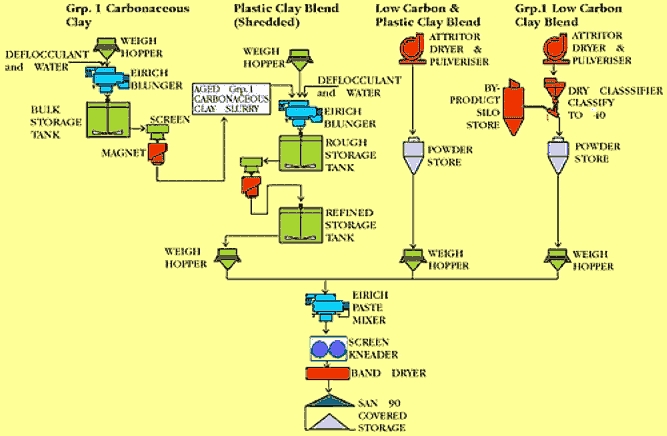 Fig. 13: Flowline for Sanblend 90 production
Fig. 13: Flowline for Sanblend 90 production
The carbonaceous clays undergo wet refining in a blunger, where water and deflocculants are added. Coarse particles are removed by screening. The low carbon and plastic clay blend is dried and powdered, and the second low carbon / plastic clay blend is dried, powdered aand air classified to remove coarse material. These three feedstocks are blended together – in carefully weighed proportions – in a crumble mixer to form a plastic mass. This is then passed through an extruder to form “noodled” shapes, which are subsequently dried in a band drier to approximately 15% moisture.
In addition, WBB also produce a range of refined ball clay blends in slurry form, in a separate dedicated plant. Computer control here facilitated a high degree of control over the proportioning and refining of multiple component blends. The main product from this plant is SanBlend 80, which uses three specially formulated feedstocks, based on a combination of North and South Devon materials. These are combined in predetermined proportions, and blunged with weighed amounts of water and deflocculants, such as sodium carbonate and sodium silicate, added from separate hoppers. Factors such as feed-rates, blunging time, discharge time, slurry density and mixer power ratings are all automatically controlled. Finally, slurry is transferred from the mixing tanks to the six main storage tanks, from which it can be pumped directly into road tankers for transportation.
6. Mineralogy and Chemistry
The dominant minerals present in ball clays are kaolinite, a micaceous mineral and quartz. Carbonaceous material and minor amounts of other minerals are often found in association with these major components. These accessory minerals can include tourmaline, feldspar, chlorite, montmorillonite, interstratified clay minerals, various titanium compounds – such as anatase, ilmenite (and occasionally rutile), and various iron minerals – such as siderite (also called chalybite), pyrites, marcasite, limonite and haematite.
For general descriptive purposes a ball clay can be regarded as an idealised ternary system consisting of kaolinite, mica and quartz. Thus, ternary composition diagrams can be prepared which show the general range of composition of Devon ball clays. Two such composition diagrams are shown in Fig.14.
In commercially available ball clays the ranges of dominant minerals are as follows:
- Kaolinite 20 – 95%
- Micaceous Mineral 5 – 45%
- Quartz 1 – 70%
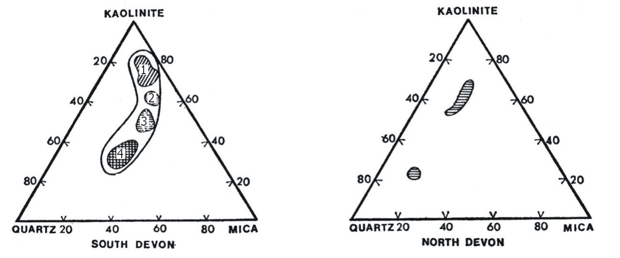 Fig. 14: Composition of Devon Ball Clays
Fig. 14: Composition of Devon Ball Clays
It may be instructive at this point to discuss in more detail the mineralogy of these three major components.
Kaolinite
The kaolinite component of the Devon ball clays is of the type often referred to as b-axis disordered kaolinite (Brindley & Robinson 1946). This type of structural disorder is readily recognised from the appearance of X- ray diffraction patterns in which the kaolinite reflections – with a k index which is not a multiple of 3 – tends to be missing or weak, whilst reflections where k=3n tend to be largely unaffected.
Various workers have suggested methods by which the degree of crystallinity of the kaolinite may be quantified. Of these methods, that accredited to Hinckley (1962) has perhaps found the widest degree of acceptance, and has in fact been used by the authors. On this scale a degree of crystallinity of zero represents completely disordered kaolinite, whilst a degree of crystallinity of approximately 2 represents nearly perfectly ordered kaolinite.
When this technique is applied to the ball clays from the South Devon deposit, a range of values from approximately 0.8 to 1.2 has been obtained, whilst the range of values obtained from clays of the North Devon deposit is much narrower – ranging from less than 0.1 to approximately 0.3. However, the derivation of numerical values of the crystallinity index is complicated by the presence of mica and quartz, and care must be exercised to obtain consistent results. Fig.15 shows X-ray diffraction traces obtained from:
(a) a sample of South Devon ball clay containing well-ordered kaolinite and
(b) a sample of North Devon ball clay containing a poorly ordered kaolinite.
The portions of the diffractogram which are particularly affected by the degree of crystallinity of the kaolinite lattice are indicated in Fig.15 by X and X1, and the Hinckley area is that designated. The number and resolution of the kaolinite peaks in these regions should be noted.
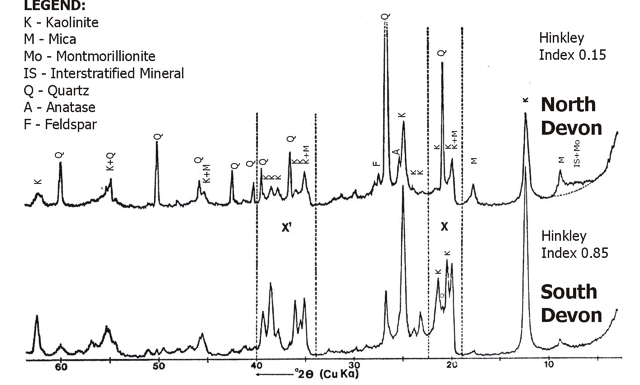
Fig. 15: X-ray diffraction traces from samples of Devon Ball Clays
In addition to the structural disorder referred to above, isomorphous substitution is known to occur within the kaolinite lattice. The most common substitution which occurs is that of iron and magnesium replacing aluminium in the octahedral layer of the kaolinite, which results in a charge imbalance which will affect certain other properties of the clay – for example, its cation exchange capacity. Upon examination of the X-ray diffraction patterns of such substituted kaolinites, certain differences are apparent when comparisons are drawn with, for example, relatively unsubstituted kaolinites, such as those associated with primary kaolins of the china clay type. These differences generally manifest themselves as broadening of the basal (001) reflections, with some tailing of the diffracted peak towards a shorter d spacing. Whilst, in part, this may be explained by the smaller grain size and lower crystallinity of the kaolinite in the ball clays, the amount of broadening generally observed is too high to be accounted for by these factors alone. Substitution within the kaolinite lattice has in fact been confirmed in a few selected clays by other techniques, notably Mossbauer spectroscopy and electron spin resonance (ESR) spectroscopy.
When ball clays are examined under the electron microscope, kaolinite crystallites can clearly be distinguished by their characteristic shape – which is generally a six-sided, (though not necessarily regular) platelet, exhibiting one or more angles of approximately 120. This external morphology, although very similar to that observed with primary kaolins, is normally less well defined – the faces and edges of the platelets being rather ragged and sometimes fractured. The incidence of twinned or multiple crystallites appears to be lower than that observed in primary kaolins. These facts are consistent with the minerals suffering attrition during transportation and sedimentation.
Micaceous Minerals
Ball clays from both the Devon deposits contain substantial amounts of micaceous minerals. Whilst, for the purpose of calculation, these minerals are often assumed to have a similar chemical composition to muscovite/paragonite, the validity of this assumption is, at best, questionable. From the broadness of the basal reflections observed on X-ray diffraction traces of most Devon ball clays, it seems probable that this micaceous mineral has a high degree of lattice substitution of iron for aluminium, and also contains a deficiency of potash, somewhat compensated for by an excess of lattice hydroxylions. It is likely that this mineral is of the type sometimes termed hydrous mica or illite.
Since this material has a very similar particle size distribution to the kaolinite with which it coexists, it has not been possible to separate a pure sample of this material and so characterise it completely from a physical and chemical point of view. But interpolation of chemical analyses of fractioned samples of ball clays certainly corresponds with the above findings.
A further complication exists with some of the South Devon ball clays, where there has been occasional evidence, again from X-ray diffraction, that there may be more than one type of micaceous mineral present. It has been suggested that these minerals, together with a small amount of detrital mica carried virtually unaltered from the Dartmoor granite mass and co-deposited with the kaolinite, may in fact be of the illite type described above.The broad peaks observed from the basal reflections of the micaceous mineral are clearly seen in the diffraction traces shown in Fig.15.
Quartz
Much of the quartz found in ball clays appears, under the optical microscope, as well-rounded grains typical of sedimentary deposits. These grains range in size from about 2 to 70 microns e.s.d. Occasionally larger corns of quartz up to 3 or 4 mm in diameter are found, and are generally rounded and sub-angular, being fairly typical of water-worn granitic quartz.
Considerable evidence exists, mainly from X-ray diffraction combined with chemical analysis, to show that the quartz which occurs in the ball clays of Devon may be considered to be a mixture of crystalline quartz and amorphous silica. It is likely that the amorphous component exists in part as a coating on the surface of the crystalline grains of quartz, as suggested by Gordon & Harris (1955) and Nagelschmidt et al (1952)), since treatment with hydrofluoric acid – which dissolves preferentially this amorphous surface layer – increases the intensity of the X-ray reflections from the quartz grains.
 Fig.16: XRD investigation in WBB Technology’s central laboratories (UK)
Fig.16: XRD investigation in WBB Technology’s central laboratories (UK)
Our own work suggests that the free silica which exists in the clay size fraction of the South Devon ball clays consists of 60% to 65% macro-crystalline quartz, with 35% to 40% of amorphous or crypto-crystalline silica; whilst in the North Devon ball clays the free silica appears to be rather less well crystallised, containing in the region of 55% to 60% of macro- crystalline quartz, with 40% to 45% of amorphous or crypto-crystalline silica. These figures may indicate that the quartz component in these ball clays has a certain amount of flint character, as typical uncalcinated flints contain of the order of 40% to 45% of macro-crystalline quartz.
Mineralogical Analysis
Whilst we may use X-ray diffraction to identify (under favourable circumstances) all the crystalline mineral phases present in the ball clay, it is more difficult to obtain quantative mineralogical data. It is relatively easy, for example, to determine the amount of quartz present in a ball clay by means of X-ray diffraction; however, it is very difficult to determine quantatively the amounts of kaolinite and micaceous mineral present. This is because the platiness of these minerals tends to make the individual crystallites orientate themselves parallel to their basal planes and perpendicular to the direction of the applied packing pressure. As a result, variations in the intensities of certain diffracted peaks occur dependent upon the degree of orientation, which is difficult to control. Various methods have been proposed to reduce this degree of orientation to a controlled and reproducible level, but at best, the results derived from such methods can only be described as semi-quantative.
It is often useful however, to have data available on the mineralogical composition of ball clays, and a method commonly employed to derive such data is the Mica Convention Rational Analysis. By this method mineralogical compositions are estimated from chemical analyses as follows:
– the soda and potash contents are used to calculate the amounts of theoretical paragonite (soda mica) and muscovite (potash mica);
– the alumina which then remains is assigned to kaolinite and any excess silica is accounted for as quartz.
This method has many obvious drawbacks, the more important of which are listed below.
1. The kaolinite is assumed to be pure A1203 2Si02 2H20. This is almost certainly not the case, especially in ball clays where substitution of iron and magnesium for aluminium probably occurs.
2. The micaceous mineral is assumed to be a mixture of ideal muscovite and ideal paragonite (K20. 3A1203. 6Si02. 2H20. and Na20. 3A1203. 6Si02. 2H20 respectively). However, hydrous mica deviates considerably from these idealised formulae.
3. The occasional presence of other aluminosilicates – for example, tourmaline, felspar, chlorites, etc. – introduces further uncertainties which can make the calculations even more invalid.
Although such criticisms of the Mica Convention Rational Analysis are very easy to make, it is difficult to find methods whereby mineralogical composition can be deduced so rapidly. However, the information derived from Rational Analysis can at least provide a guide to mineralogical composition and may suggest reasons for differences in the physical properties and performance of the materials in ceramic systems.
 Fig. 17: Electron micrsoscopy in WBB Technology’s central research laboratories
Fig. 17: Electron micrsoscopy in WBB Technology’s central research laboratories
A rather more sophisticated type of mineralogical analysis has been described by West (1970), which attempts to account for all the minerals which may be present in a clay and whilst we have applied this method with some success to china clays where the chemical compositions of the kaolinite and mica are known with more certainty, we have had less success in applying it to ball clays.
 Fig. 18: Electron micrograph of ball clay
Fig. 18: Electron micrograph of ball clay
7. Accessory Minerals
Ball clays contain varying amounts of several accessory minerals. It is worth while considering a few of the more important of these separately and indicating the effects that these may have upon the use of the ball clays.
Carbonaceous Matter
From the point of view of its widespread occurrence, its occasional existence in high quantities, and the far-reaching effects its presence has in modifying certain important physical properties of the ball clays, carbonaceous matter is possibly the most significant contaminant mineral found associated with the ball clays.
This particular mineral occurs in ball clays in two distinct physical forms. The first is a lump form generally termed lignite, much of which is greater than 50 microns e.s.d., which is usually removed from the ceramic system by screening. The second is a colloidal form which exists as a coating on the individual crystallites of the clay mineral. The presence of either form is often indicated by a dark colouration imparted to the clay – the density of the colouring being such that some clays are black in the crude form; hence the origin of the term black ball clay. Broadly, the density of the colouring is proportional to the amount of carbonaceous matter present.
Various methods exist for the estimation of the amount of carbonaceous matter present in ball clays. The Mica Convention Rational Analysis, referred to previously, does give an estimation of the carbonaceous matter present, and generally this compares reasonably well with values obtained by direct measurement. The carbonaceous matter may be determined directly by various chemical and physical methods – for example, by combustion in a carbon train; by thermogravimetric analysis; or by various oxidation techniques such as the Walkley & Black method (1934), which involves the oxidation of the carbonaceous matter by potassium dichromate in the presence of concentrated sulphuric acid; or by Tidys method using potassium permanganate or hydrogen peroxide for the oxidation.
The composition of lignite has been extensively studied, notably by Worrall et al (1953), and the principal constituents of lignite are now considered to be: Resins and tannins, Fats and waxes, Lignin and humic acids, Cellulose and some residual mineral matter.
An analysis of a typical sample of lignite is given below:
Carbon 46.8%; Hydrogen 3.5%; Nitrogen 0.5%; Sulphur 3.2%; Oxygen 18.3%; Ash 27.8%.
Because of the difficulty in obtaining suitable samples, most of the studies of carbonaceous matter have been undertaken on the coarse lignite fraction which, generally speaking, is of least interest to the practising ceramist. Much of the modification to the physical properties of the ball clay which occurs is believed to be due to the presence of the finely dispersed collodial material.
The collodial carbonaceous material has been loosely termed humic acid and has been shown by various workers to consist of a heterogeneous mixture of many compounds for which no single structural formula will suffice.
It is known that the humic acids have large cation exchange capacities, often of the order of 300 milli-equivalents per 100 grammes, and this can result in strong buffering action and the production of extremely stable deflocculated water slips of very high fluidity. This is illustrated in Fig.19 where the deflocculation characteristics of a highly carbonaceous clay are shown, together with those of a low carbon clay of an otherwise similar mineralogical make-up.
Apart from these desirable and useful effects on fluid properties; carbonaceous matter – particularly humic acid – has a profound effect upon the green strength of clays. This is illustrated in Fig.20 where the moduli of rupture of a series of ball clays have been plotted against carbonaceous matter content. These clays were specifically selected as being of a similar mineralogy and occuring in a geologically similar environment.
The main disadvantage of particulate carbonaceous matter in relation to the ceramic system is that if it is present in any quantity – say above 1% to 1.5% – then great care is needed in the firing process in order to avoid problems with, for example, black coring or pin holing. Carbonaceous matter can also contribute to the problem of specking of ware, largely due to the relatively high amounts of ferruginous matter associated with the rather coarsely particulate (approximately 50 microns e.s.d.) lignite.
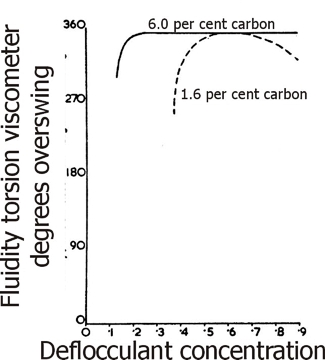
Fig. 19: Deflocculation characteristics of a highly carbonaceous clay
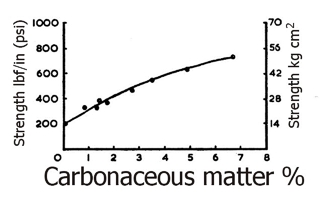
Fig. 20: Moduli of rupture of a series of ball clays plotted against carbonaceous matter content
Montmorillonite
Montmorillonite can be considered as the second most influential impurity in ball clays. Unfortunately it is an extremely difficult mineral to detect when present in small quantities and, as such, the presence of montmorillonite is often inferred from changes in the properties of clays. Detection of montmorillonite is usually made by X-ray diffraction on oriented specimens. However, montmorillonite is a very poor reflector of X-rays and, even under favourable conditions, it is difficult to positively identify this mineral when it occurs in levels less than 5%. But often minor changes in the X-ray diffraction pattern can be seen which makes possible the inference of quantities as low as 2% to 3%.
Positive identification of this mineral is possible when it occurs at levels of the order of 5% and above, and this is done by re-examining the specimen after treatment with a suitable polyhydric alcohol, usually ethylene glycol, which replaces the water solvating the interlayer cations and causes interlayer expansion. This results in a movement of the basal reflections, thus making identification positive. At lower levels of concentration electron microscopy may also be used as a means of identification of montmorillonite, but it is difficult to obtain quantative data. It will be seen later that levels of montmorillonite well below 5% are sufficient to have a marked effect on many of the physical properties of the clays.
The effect of montmorillonite on ball clays is to increase the green strength, the plasticity and the dry contraction. Table 1 illustrates these effects and also summarises experiments in which successive amounts of a commercial bentonite were added to a siliceous ball clay.
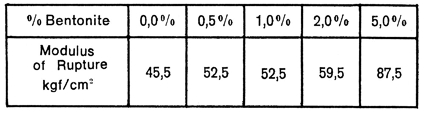 Table I
Table I
In the firing of clays, the presence of montmorillonite increases the firing contraction and, because it invariably contains iron, also has a deleterious effect on the fired colour of the product.
Probably the major effect of montmorillonite however, is in ceramic casting slips, where it materially affects the deflocculation behaviour of the clay. This results in a high deflocculant demand, low slip stability and high slip viscosity. The slips also tend to thicken markedly on ageing.
Interstratified Clay Minerals
It has been suggested that montmorillonites have been formed from the breakdown and weathering of the micaceous compounds in the country rock under alkaline conditions. Various intermediate stages in this formation have been suggested, including vermiculite, illite and mixed layer intergrowths of both.
Whilst the detection of interstratified minerals is at least as difficult as the detection of montmorillonite, and possibly more so, evidence has been found in some of the North Devon clays to suggest the presence of mixed layer illite/montmorillonite. Some indirect evidence for the occurrence of small quantities of interlayer minerals in the South Devon clays has been reported.
Little work has been done to discover the exact effects of interlayer minerals upon the physical properties of ball clays for a variety of reasons, but it is reasonable to assume that they would probably be similar to the modifications imposed by the presence of montmorillonite – namely increase in green strength; increase in plasticity; increase in deflocculant demand; increase in slip viscosity; lowering of slip stability; etc. It is likely however that interlayer minerals are not quite as potent as montmorillonite in this respect.
Iron Compounds
Certain specific iron compounds are occasionally present in ball clays. These are usually marcasite (FeS2), or pyrites (FeS2) and siderite (FeC03). Iron is also associated with the organic matter.
Very little of the free iron which occurs naturally in the ball clays (probably of the order of 0.2% to 0.4% as Fe203) is accounted for by the above-mentioned impurities, the majority being present as a hydrated oxide – e.g. limonite (Fe203 nH20) – which occurs as a coating on the individual crystallites of kaolinite, mica and quartz. The composition of the iron compounds is very variable, resulting in a varying degree of colour, dependent upon the amount of hydration and the fineness of the material. The iron also occurs as an amorphous substituent within the lattice of the kaolinite and the mica.
The principal effect of nodular iron contaminants within clays is on the fired colour, where it results in specking – the specks produced being red through brown to black, according to the intensity and type of the iron contaminant and the firing conditions. The isomorphously substituted iron present in the mineral lattice does not give rise to specking on firing, but is, to some extent, responsible for background colour which may be cream or buff depending on the level present.
Titania
The average level of Ti02 in Devon ball clays is about 1%, although levels as high as 2.5% have been observed. The principal form in which the titania occurs is as anatase, the presence of which may be detected by X-ray diffraction. One of the anatase lines used for identification is in fact shown on the X-ray diffraction trace in Fig.8. Rutile, a second isomorphous form of titania, is found on very rare occasions.
The main effect of titania is on fired colour, where it is usually classified with the iron and, if the total of Fe203 + Ti02 exceeds 2.5%, the clay will normally fire to a cream/dark colour at temperatures around 12000C.
Soluble Salts
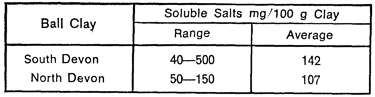 Table II
Table II
Although soluble salts may not arise from one specific mineral, they are commonly found as a contaminant in virtually all ball clays. The presence of excessive amounts of such salts can give rise to problems in the ceramic industry, such as scumming, which later results in certain glaze defects, like edge-peeling. In slip casting operations the amount of soluble sulphate present can give rise to variability in the properties of the casting slip produced.
The range of total soluble salts varies considerably from clay to clay and from area to area. Some values given by Holdridge (1956) are summarised in Table 2.
8. Classification of Ball Clays
a. South Devon Clays
For practical purposes it is convenient to divide clays of the South Devon area into four distinct groups. The original dilatometric basis for this has been described in detail by Mitchell (1963) and, broadly speaking, this method classifies the clays according to their mineralogical composition. The four groups may be described generally as follows:
Group 1
Clays in this group are noted for their excellent fired colour. They are dominantly kaolinitic with low mica and quartz contents and frequently contain appreciable quantities of carbonaceous matter. This carbonaceous matter can act as a protective colloid in the production of clay/water slips, and ensures a high fluidity and low alkali demand at optimum deflocculation. Historically, clays from this group were termed black ball clays.
Group 2
The clays from this group, the so-called dark blue clays, are again dominantly kaolinitic, but contain rather more micaceous mineral and quartz than the clays of Group 1. These clays also contain a certain amount of carbonaceous matter which is generally helpful in producing easily deflocculated casting slips. The clays of this group are amongst the strongest clays found in the South Devon deposit.
Group 3
The clays of this group, the so-called light blue clays, contain rather less kaolinite and higher proportions of mica and quartz than clays of the preceding two groups. Generally speaking, the carbon content of these clays is low (less than 0.5%). Clays in this group tend to be extremely thixotropic and use is made of this property in the production of blended clays intended for slip casting.
The clays of this group are the most siliceous of the South Devon clays. They are generally similar to the clays of Group 3, but with higher quartz contents and lower carbonaceous contents.
Fig.21 shows the general form of the dilatometric curves of clays of each of these groups – the original basis of this classification. To a certain extent these groupings are arbitrary and, inevitably, some overlap occurs with some seams falling between adjacent groups.
2. North Devon Clays
To draw up a classification for clays from the North Devon deposits as generalised as that drawn up for the South Devon clays is more difficult, but basically there are two distinct types of clay to be found in North Devon.
The basis of this distinction centres around differences observed in some of the rheological parameters of these clays, which are believed to arise through small changes in their mineralogy. Although both basic types of clay are predominantly kaolinitic, clays of Type 1 contain, in addition, appreciable quantities of montmorillonite, resulting in high deflocculant demand values and relatively high slip viscosities. Clays of Type 2, however, contain relatively little montmorillonite, and consequently have lower deflocculant demand values and lower slip viscosities.
To some extent the presence of montmorillonite in the North Devon clays is the reason for their inherent high strength and plasticity. It appears, however, that the montmorillonite is often present as a coating on the kaolinite crystallites, rather than as a physically distinct crystal phase. For this reason excessive drying shrinkages are not observed.
This relatively simple classification can, however, be confused somewhat by the presence of extremely siliceous types of clay found in association with both the above groups.
Table 3 summarises the chemical analyses of clays typical of the above- mentioned groups of South Devon and North Devon clays.
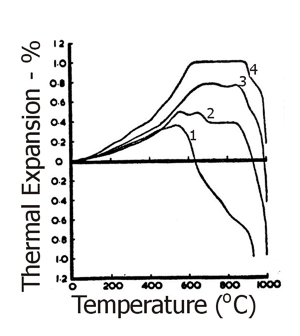 Fig. 21: Characteristic thermal expansions of South Devon Ball Clays
Fig. 21: Characteristic thermal expansions of South Devon Ball Clays
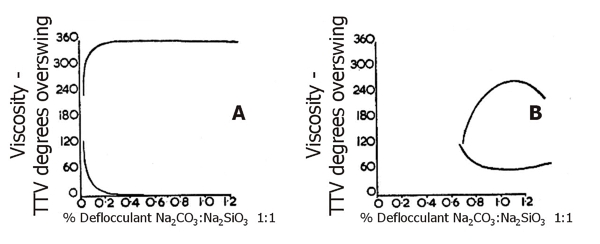 Fig. 22: Deflocculation characteristics of English Ball Clays
Fig. 22: Deflocculation characteristics of English Ball Clays
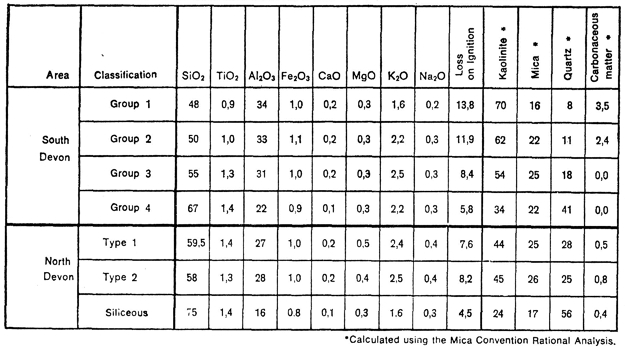 Table 3: Mean Chemical and Mineralogical analyses of Typical Devon Ball Clays
Table 3: Mean Chemical and Mineralogical analyses of Typical Devon Ball Clays
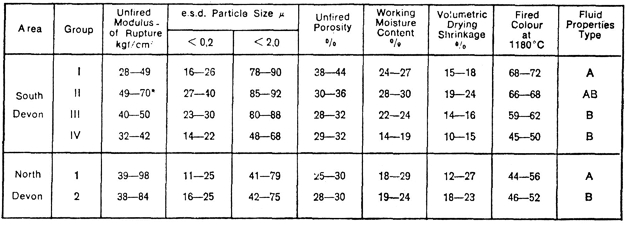 Table 4: Physical Properties of Devon Ball Clays
Table 4: Physical Properties of Devon Ball Clays
9. Physical Properties of Ball Clays
The principal physical characteristics of ball clays from the various production areas in Devon are summarised in Table 4. Generally, the widest spectrum of properties is obtained from the South Devon clays, this being consistent with their origins and previously described mineralogy.
Fig.22 illustrates typical ball clay deflocculation curves of Type A and Type B referred to in Table 4. Figs. 23 and 24 illustrate fired colour and vitrification characteristics of the four main groups of South Devon clays.
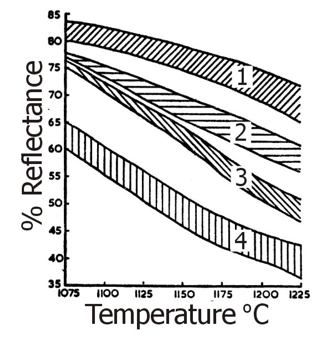 Fig 23: Fired colour / Temperature Relationship of South Devon Ball Clays
Fig 23: Fired colour / Temperature Relationship of South Devon Ball Clays
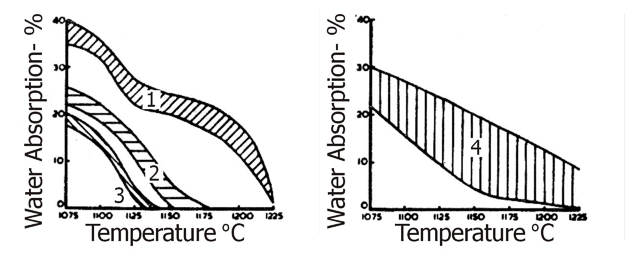 Fig 24: Vitrification characteristics of South Devon Ball Clays.
Fig 24: Vitrification characteristics of South Devon Ball Clays.
It is rare that any single clay can be found which completely satisfies all the manufacturing requirements of any one product. For this reason blended clays are produced which consist of many components from several individual seams, each selected in order to provide its own specific contribution to the physical properties of the final blend. Thus, it is possible to buy blends of South Devon Group 1, Group 2 and Group 3 clays, as well as blended clays containing components from the North Devon production area.
Obviously different sets of criteria are important to the production of any specific blend. For example, a blended clay produced principally for use in the sanitaryware industry would have as its principal requirement an easily deflocculated, easily controlled, fast casting, stable, predictable slip, with high green strength, yet without excessive drying contraction which can result in cracking and distortion. Such a blend may be produced by the judicious mixing of the carbonaceous Group 1 South Devon clays with, for example, plastic Group 2 clays and some of the North Devon materials.
On the other hand, a ball clay for use in the electrical porcelain industry would need to be highly plastic and would usually contain finely dispersed micaceous material to assist in vitrification. Undoubtedly, the most suitable clays for the production of this type of ware are blends of the North Devon clays. These have the additional advantage of remaining workable over a wide range of moisture content and are also eminently suitable for extrusion of large pieces which are to be subsequently finished by turning.
The ceramic industry has its own set of criteria for the ball clays it uses in its formulations, and even different manufacturers in the same sphere of operation may have slightly different requirements, even of the same material. Consequently, the ball clay producer, ever mindful of the changing circumstances throughout the industry due to the rapid advancement of technology, continues to improve the flexibility and control of each blend he produces, and additionally continues his researches for new blends to remain abreast of these steady improvements and modifications in production methods.
(c) 1984 Watts Blake Bearne & Co PLC
References
1. SCOTT, A. 1929. Ball Clays. Memoirs of the Geological Survey,Sp.Report on the Mineral Resources of G.B.
2. BRISTOW, C.M. 1968. The derivation of the Tertiary sediments in the Petrockstow Basin, North Devon, Proceedings of the Ussher Society 2, p.2935
3. BROOKS, M. and JAMES, D.G. 1975 The geological results of seismic refraction surveys in the Bristol Channel, 197073, Journal of the Geological Society 131, p.163182
4. FLETCHER, B.N. 1975 A new Tertiary basin east of Lundy Island. Journal of the Geological Society 131, p.223225
5. BRISTOW, C.M. and HUGHES, D.E. 1971 A Tertiary thrust fault on the southern margin of the Bovey Basin, Geological Magazine, 108, p.6167
6. BRISTOW, C.,M. et al. 1992 Palaeogene basin development: new evidence from the Southern Petrockstow Basin, Devon, Proceedings of the Ussher Society, 8, p1922
7. EDWARDS, R.A. 1976. Tertiary sediments and structure of the Bovey Basin, South Devon. Proceedings of the Geologists Association, 87, p.126
8. HOLLOWAY, S. and CHADWICK, R.A. 1986. The SticklepathLustleigh fault zone: Tertiary sinistral reactivation of a Variscan dextral strikeslip fault. Journal of the Geological Society 143, p.447452
9. FASHAM, M.J.R. 1971. A gravity survey of the Bovey Basin, Devon. Geological Magazine 108, p.119130
10. FRESHNEY E.C. et al. 1979. Geology of the country around Chumleigh. Memoirs of the Geological Society of Great Britain, London.
11. CHANDLER, M.E.J. 1957. The Oligocene flora of the Bovey Tracey lake basin, Devonshire. Bulletin of the British Museum (Natural History), 3, p.71123
12. CHANDLER, M.E.J. 1964. The Lower Tertiary floras of Southern England, Part 4. British Museum (Natural History), London.
13. VINCENT, A. 1974. Sedimentary Environments of the Bovey Basin. Unpublished M.Phil. Thesis, Univ. of Surrey.
14. BRINDLEY, G.W. & K. ROBINSON. 1946. Randomness in the Structures of Kaolinitic Clay Minerals. Trans. Faraday Soc., 42B, p.198.
15. HINCKLEY, D.N. 1962. Variability in Crystallinity Values Among the Kaolin Deposits of the Coastal Plain of Georgia & South Carolina. Proc. 11th Nat.Conf. on Clays & Clay Mins. p.229.
16. MALDEN, P.J. & R.E. MEADS. 1967. Substitution by Iron in Kaolinite. Nature, Lond., 215, p.844.
17. ANGEL, B.R. & P.L. HALL. 1972. Electron Spin Resonance of Kaolins. Proc.Int.Clay Conf.,Madrid. p.47.
18. BROWN, G. & K. NORRISH. 1952. Hydrous Micas. Min. Mag, 29, p929.
19. GRIM, R.E. et al. 1937. Mica in Argillaceous Sediments. Amer.Min. 22, p.813.
20. GORDON,R.L. & G.W. HARRIS. 1955. Effect of Particle Size on the Quantitative Determination of Quartz. Nature, Lond., 175, p.1135.
21. NAGELSCHMIDT, G. et al. 1952. Surface of Finely Ground Silica. Nature, Lond., 169, p539.
22. BRINDLEY, G.W. & S.S. KURTOSSY. 1956. Quantitative Determination of Kaolinite by XRay Diffraction. Amer.M
23. NISKANEN, E. 1964. Reduction of Orientation Effects in the Quantitative XRay Diffraction Analysis of Kaolin Minerals.Amer.Min., 49, p.705.
24. WEST, R. 1970. Estimation of Constitution of Whiteware Clays Using the Computer. Proc. Fall Meeting, Amer. Cer. Soc., Material & Equipment & Whitewares Divn.
25. WALKLEY, A. & I. ARMSTRONG BLACK. 1934. An Examination of the Degtjareff Method for Determining Soil Organic Matter, and a Proposed Modification of the Chromic Acid Titration Method. Soil Science, 37, p.29.
26. WORRALL, W.E. & C.V. GREEN. 1953. The Organic Matter in Ball Clays. Trans.Brit.Cer.Soc., 52, p.528.
27. WORRALL, W.E. 1956. The Organic Matter in Clays. Trans. Brit. Cer.Soc., 55,p.689.
28. STEVENSON, F.J. & J.H.A. BUTLER. 1969. Organic Geochemistry (Editors: Eglington & Murphy). Longmans, Green & Co., Lond.
29. ALTSCHULER, Z.S. & E.J. DWORNIK. 1963. Transformation of Montmorillonite to Kaolinite During Weathering. Science, 141, p.148.
30. BEST, R. & FOOKES, P.G. 1970. Some Geotechnical and Sedimentary Aspects of Ball Clays from Devon. Q.J.Eng.Geol.3, p.207
31. HOLDRIDGE, D.A. 1956. Ball Clays & Their Properties. Trans.Brit.Cer.Soc., 65, p.369.
32. MITCHELL, D. 1963. Ball Clays & Their Properties. Proc. Norsk. Keramisk Selskap, Spring Meeting p.47.